ORIGINAL RESEARCH ARTICLE
Prevalence of Gastrointestinal Parasites in Cats and Dogs on Intake to Northern Mississippi Shelters
Dallas E. Riley*, W. Cooper Brookshire, Philip A. Bushby, T. Graham Rosser and Jacob M. Shivley
Mississippi State University College of Veterinary Medicine, Starkville, MS, USA
Abstract
Animal shelters frequently administer multiple anthelminthic therapies for gastrointestinal parasites to animals on intake, but superfluous use of anthelmintics may contribute to resistance over time. Literature results for gastrointestinal parasite prevalence vary by region. This study’s objective was to evaluate parasite prevalence in animals upon intake to Mississippi shelters and determine risk factors for parasitism. Fecal samples were obtained from cats and dogs and examined via passive floatation. Presence or absence of common parasite eggs was found along with descriptive characteristics of the animal. Multivariable, multilevel logistic regression (SAS9.4) was performed to test for associations (alpha = 0.05). Prevalence for overall parasitism in cats and dogs was 48% (95% confidence interval [CI]: 38.9%–57.2%) and 74% (95% CI: 69.6%–77.3%), respectively. Age and intake type were found to have significant associations with parasitism in cats (P = 0.027, P = 0.015). Age and sex were observed to have significant associations with parasitism in dogs (P = 0.035, P ≤ 0.0001). These findings suggest implications for targeted empirical anthelmintic therapy.
Keywords: Parasite; parasites; cat; dog; canine; feline; shelter; intake; pyrantel; strongid
Citation: Journal of Shelter Medicine and Community Animal Health 2026, 5: 160 - http://dx.doi.org/10.56771/jsmcah.v5.160
Copyright: © 2026 D.E. Riley et al. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material for any purpose, even commercially, provided the original work is properly cited and states its license.
Received: 24 September 2025; Revised: 22 December 2025; Accepted: 24 December 2025; Published: 27 February 2026
Competing interests and funding: The authors declare that they have no conflicts of interest to disclose. This work was supported by Mississippi State University College of Veterinary Medicine Office of Research and Graduate Studies House Officer Grant and the National Institute of General Medical Sciences of the National Institutes of Health (Award Number U54GM115428).
Correspondence: *Dallas E. Riley, Mississippi State University College of Veterinary Medicine, Department of Clinical Sciences 240 Wise Center Dr Mississippi State, MS 39762. Email: dallas.riley@msstate.edu
Reviewers: Heather Walden, Adebayo Molehin
An animal’s shelter intake examination is the initial assessment of general health and the first opportunity for medical intervention; occasionally, it may be the only opportunity for medical intervention. Many organizations utilize general intake protocols, which often include vaccinations, flea and tick preventatives, and deworming treatments. Based on shelter resource availability and staff training, diagnostics such as fecal direct smear or floatation, Wood’s lamp screening, heartworm antigen testing may not be practically performed on every animal. By definition,1 this empirical provision of care is based on previous experience and perceived conditions rather than each animal’s confirmed morbidities or individual risk for threats to health. Resources such as staff availability, staff education and training, diagnostic tools, and financial support contribute to decisions made during the intake process and throughout an animal’s length of stay. A single protocol does not necessarily apply to all animals or meet individual needs. It must be acknowledged that greater contributions of time and attention to detail are necessary to individualize shelter animal care and that intake protocols simplify general care for the population. Nevertheless, empirical treatment such as deworming poses the opportunity to create more long-term harm than short-term benefit. Physically, animals may have parasitism that is not susceptible to treatments due to latency, incubation period, or incorrect diagnosis and therapy. Mentally, animals are subjected to additional handling and environmental stress. Financially and regarding staff allocation of time, the shelter utilizes resources that were potentially unnecessary and may have been valuable for other uses. Regarding specifically anthelmintic therapies, administration of superfluous therapy may contribute to development of anthelmintic resistance.
It is beneficial to evaluate regional prevalence of gastrointestinal parasites and establish risk factors for parasitism to guide empirical protocols. When discussing internal and external parasite treatment, the Association of Shelter Veterinarians (ASV) Guidelines for Standards of Care in Animal Shelters2 (p. 32) confer that geographical risks and the impacts of particular parasites on human and animal health should be considered when creating a parasite control program. Previous literature has indicated a higher prevalence of common gastrointestinal parasites in dogs of southern regions of the United States compared to other regions.3–6 Additionally, young age and neuter status have been identified as risk factors in dogs.3,5,6 These studies assess pets or animals already housed in shelters, animals that would be more likely to have undergone intake procedures; therefore, these studies may not give an accurate picture of an animal’s risk for having particular parasites upon entry to the shelter, limiting their value in guiding decisions specifically for animal intake. In the interest of gauging public health risks, Campanale et al.7 evaluated intake and discharge fecal examinations to estimate prevalence of zoonotic parasites in dogs at a north central Florida animal shelter. The authors found that 54% of dogs were diagnosed with gastrointestinal parasites at admission, with Ancylostoma spp. (hookworms) and Toxocara canis (roundworms) being most common. Because of regional proximity and similar climate effects, the high prevalence of parasites and zoonotic threat to staff can potentially be extrapolated to Mississippi shelters. Congruent with regional proximity, Hoggard et al.4 sampled cats in a Georgia shelter that had not been treated with an endoparasiticide and found a prevalence of 45.6%, with Toxocara cati described as the most prevalent parasite. Prevalence data for cats in the United States are primarily regionally reported, and it is likely that difficulty in obtaining samples prior to antiparasitic therapy creates a challenge in reporting these data. Additionally, evidence regarding risk factors for parasitism in shelter cats is greatly limited in published literature. The Companion Animal Parasite Council (CAPC) is a commonly cited source that reports annual prevalence data for cats and dogs across the nation. Reports for Mississippi in the last 5 years (2018–2023) have all been low (<10% regardless of species or internal parasite).8,9,10 Annual reports are reliant on case submission, and cases are most frequently sourced from owned animals; therefore, these data likely cannot be relied upon for decision-making in shelters.
The objective of this study is to estimate the prevalence of common gastrointestinal parasites in animals entering northern Mississippi shelters and evaluate risk factors for parasitism. The obtained data will determine if Mississippi regional data are consistent with other literature and expand knowledge of prevalence and risk factors for cats and dogs entering Mississippi shelters. This information could potentially be extrapolated to other southern areas of the United States and will be vital to improve meeting the needs of shelter animals and mitigating public health risks. The aims of anthelmintic therapy are to alleviate clinical severity in the patient, reduce environmental contamination, and diminish zoonotic risk. To provide effective care, common parasites affecting the regional population and individual animal risk should be considered when creating intake protocols or administering population-level therapy.
Materials and methods
Sample size calculation
To reliably estimate regional prevalence, sample size estimations for cats and dogs were calculated using a cross-sectional studies calculator within Epi InfoTM (Version 7.2.6.0, Centers for Disease Control and Prevention).11 A power of 80% and desired 95% confidence interval were applied. The sample size estimation for dogs was determined based on previously reported differences in prevalence of nematodes by categorization of age. It was estimated that 250 dogs would be required to note differences between age groups. Alternative sample size calculations were made based on prevalence publications for intake type and sex; however, the age variable resulted in the largest sample size required to establish differences. Similarly, the sample size estimation for cats was determined using published differences in the prevalence of coccidia by categorization of age. An estimation of approximately 160 cats would be required to note differences between age groups. Due to lack of published prevalences for other variables (intake type, sex, etc.), this estimation was maintained for sample goals.
Sampling and evaluation
All procedures within this study were performed with consent from shelter management and were approved under the guidelines set by the Institutional Animal Care and Use Committee of Mississippi State University College of Veterinary Medicine.
Fresh fecal samples were obtained from cats and dogs entering three northern Mississippi shelters and cats presenting for ‘trap, neuter, return’ (TNR) surgical sterilization at a high quality high volume spay neuter clinic. Sample collection began in January 2022 and concluded in November 2023. Feces were obtained during animal intake procedures prior to the administration of any antiparasitic drugs via environmental collection or fecal loop sampling. Feces were removed from the enclosure if present and the animal was singly housed; if multiple animals inhabited an enclosure or no feces were present at the time of visitation, feces were obtained via fecal loop sampling. The scheduled times of visitation coupled with routine shelter cleaning protocols ensured that all feces obtained from kennels were at most 4 h old. All TNR cats were sampled with a fecal loop while under general anesthesia for routine procedures not related to this study.
The sample population excluded animals with previous administration of oral anthelmintics, topical antiparasitics, or preventive adornments such as flea and tick collars. All TNR cats were assumed to have never received any antiparasitic therapies prior to presentation. Additionally, animals younger than 12 weeks of age were excluded from fecal loop sampling to alleviate sampling challenges in small patients and reduce the handling of the shelter’s most disease susceptible individuals. Environmental samples from animals younger than 12 weeks of age were collected and utilized in this study.
Descriptive information about each study patient was catalogued with each sample. Information such as animal identification, shelter, species, age, sex, and intake type were documented. Age was parsed into four categories: pediatric (less than 6 months of age), juvenile (6 months to 1 year), adult (1–7 years), and senior (greater than 7 years). All animals were aged by the primary investigator via tooth eruption and phenotypic characteristics (e.g. greying fur around the face or cataracts).12,13 Lastly, intake type was documented according to the receiving shelter’s definition. For example, an animal was deemed a ‘Transfer’ if the shelter in which it was sampled received the animal from another shelter. This particular intake type was included for study analysis to account for common transfer of animals between northern Mississippi shelters. Transfer animals are originally taken in by one shelter and transported to another shelter that is more capable of caring for the animal’s particular needs. These animals may undergo intake procedures at the receiving shelter regardless of care provided by the previous shelter.
All samples were refrigerated upon collection and evaluated within 48 h. Passive fecal floatation with a commercially available sodium nitrate solution (Fecasol®, specific gravity of 1.200) was performed. In lieu of centrifugation, passive floatation rested for 15 min to allow for optimal rising of ova to the surface.14,15,16 Samples were weighed using a calibrated analytical balance. When a sample contained less than two grams of feces, the entirety of the sample was utilized in floatation; if a sample contained over two grams of feces, only two grams were utilized. Each sample (ranging between 0.1 and 2 grams of feces) was floated with 15 ml of Fecasol®. Each sample was assessed for the presence of presumed Ancylostoma spp., Toxocara spp., Trichuris spp. and Cystoisospora spp. via visual assessment in alignment with published gastrointestinal parasite keys16,17 (p. 364–366, 371–372); species identification was not performed. Though other incidental parasites were observed, these data were not included in the statistical analysis. Fecal egg counts were not assessed and the data were catalogued as ‘present’ or ‘absent’ for analysis.
Analysis
Data were analyzed using Version 2312 of Microsoft Excel18 and Version 9.4 of the Statistical Analysis System (SAS) for Windows.19 Descriptive statistics were documented by species. Prevalence by species was determined for overall parasitism, specific parasites (Ancylostoma spp., Toxocara spp., Trichuris spp., and Cystoisospora spp.), and coinfections, along with Clopper-Pearson 95% confidence intervals. For statistical analysis, multivariable logistic regression with manual forward selection (PROC LOGISTIC) was performed to test for risk factors; variables (shelter, age, sex, intake type) were entered into the model in order of smallest P-value until no significant variables were left. Lastly, the interaction of sex and age was tested with individual sex and age associations. Model validation was performed with the Hosmer–Lemeshow test and residuals were visually inspected. An alpha of 0.05 was used for all analyses.
Results
Feline
Descriptive categorical data are summarized in Table 1. The mean age of cats sampled was 16 months; the youngest and oldest cats were 6 weeks and 12 years, respectively. Five different intake types were documented: stray, owner surrender, TNR, transfer, and unknown. Prevalence for overall parasitism (presence of any one of the evaluated common gastrointestinal parasites) in cats was 48% (95% confidence interval [CI]: 38.9%–57.2%), and 46% (95% CI: 36.5%–54.8%) of all cats posed a zoonotic risk to humans. Toxocara spp. and Ancylostoma spp. were equally prevalent among the samples at 28.0% (95% CI: 20.0%–36.4%, 20.7%–37.3%, respectively). Cystoisospora spp. demonstrated the lowest prevalence among study animals at 9.0% (95% CI: 4.5%–15.4%). Figure 1 illustrates the prevalence of each parasite compared to overall parasitism and samples with no parasites seen. Other parasites observed infrequently included Spirometra spp. eggs, Demodex spp. mites, and tapeworm ova of unknown species. Approximately, 15% (95% CI: 6.8%–30.7%) of positive samples contained multiple parasites, suggesting that concurrent infections were relatively uncommon compared to infections of a singular parasite type.
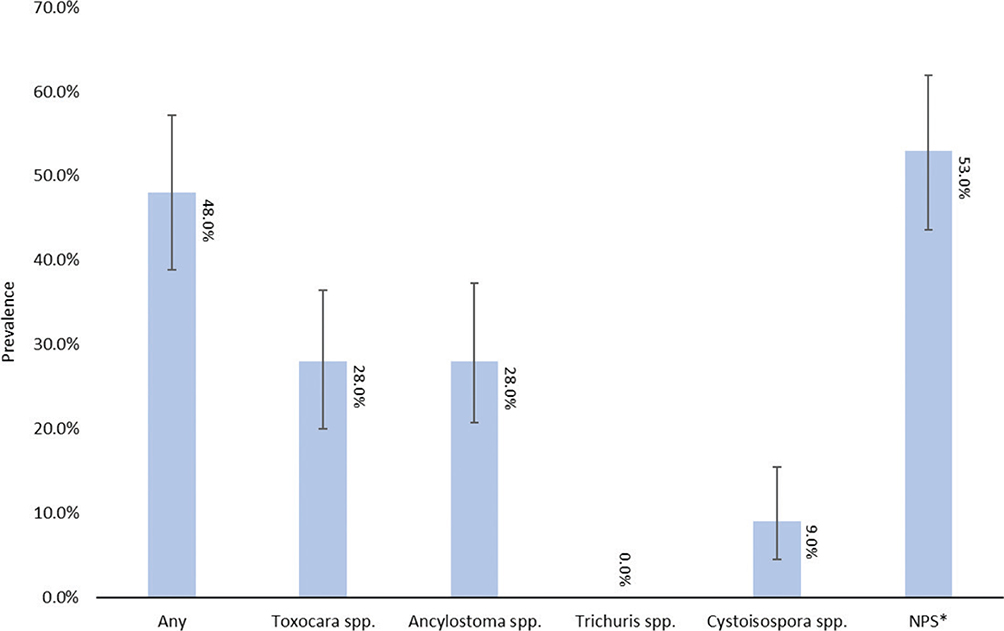
Fig. 1. Gastrointestinal parasite prevalence in feline fecal samples.
*NPS= No Parasites Seen; Error bars denote 95% confidence intervals.
| Raw count of sampled cats by descriptive characteristic and parasitism status* | |||
| Parasitized | Total count | ||
| Yes | No | ||
| Age category | |||
| Pediatric (< 6 months) | 20 | 25 | 45 |
| Juvenile (6 months – 1 year) | 5 | 2 | 7 |
| Adult (1 year – 7 years) | 29 | 33 | 62 |
| Senior (> 7 years) | 1 | 3 | 4 |
| Unknown | 4 | 1 | 5 |
| Sex | |||
| Male | 29 | 27 | 56 |
| Female | 29 | 36 | 65 |
| Unknown | 1 | 1 | 2 |
| Altered status | |||
| Intact | 57 | 54 | 111 |
| Altered | 0 | 9 | 9 |
| Unknown | 2 | 1 | 3 |
| Intake source | |||
| Stray | 29 | 34 | 63 |
| Owner surrender | 5 | 14 | 19 |
| Transfer | 21 | 9 | 30 |
| TNR | 3 | 4 | 7 |
| Unknown | 2 | 2 | 4 |
| *Counts indicate presence of at least one of the common gastrointestinal parasites evaluated in this study (roundworms, hookworms, whipworms, coccidia). | |||
Logistic regression was used to identify risk factors for parasitism. Strays had 4.4 times greater odds of parasitism over other intake types (P = 0.014). Age was associated with specifically Ancylostoma spp. infections. Juvenile cats had 13.6 times greater odds over pediatric and 4.9 times greater odds over adult cats of presenting to a shelter with hookworms (P = 0.027, P = 0.027, respectively); conversely, pediatric cats had approximately half the odds of presenting with Ancylostoma spp. compared to adult cats (P = 0.027).
Canine
Descriptive categorical data are summarized in Table 2. The mean age of dogs sampled was 21 months; the youngest and oldest dogs sampled were 6 weeks and 10 years, respectively. Five different intake types were documented: stray, owner surrender, seized, transfer, and unknown. Prevalence for overall parasitism for all dogs was 74% (95% CI: 69.6%–77.3%), and 68% (95% CI: 64.0%–68.2%) of all dogs posed a zoonotic risk to humans (Fig. 2). Ancylostoma spp. were the most commonly observed parasite among the samples at 62.0% (95% CI: 57.6%–66.1%) prevalence. Cystoisospora spp. demonstrated the lowest prevalence among study patients at 4.0% (95% CI: 2.5%–6.1%). Other parasites observed infrequently included Demodex spp. mites and tapeworm ova of unknown species. Concurrent infections were more common in dogs than cats. Approximately, 28% (95% CI: 22%–29%) of positive samples contained more than one parasite, though singular parasite infections were most common overall. One dog was observed to have all four evaluated common gastrointestinal parasites.
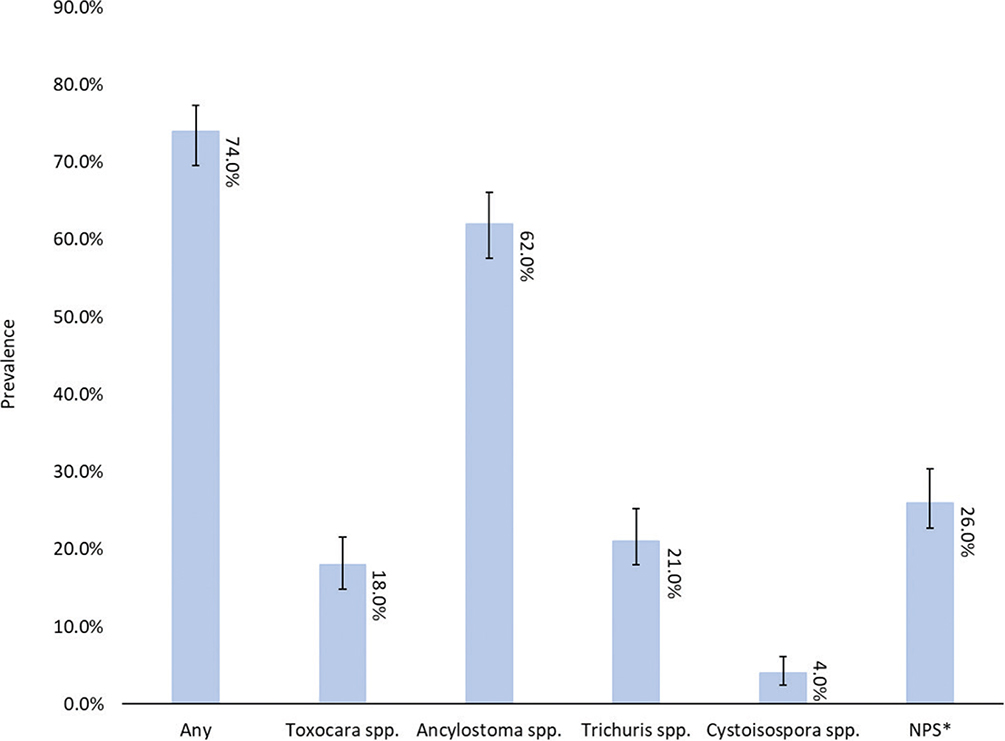
Fig. 2. Gastrointestinal parasite prevalence in canine fecal samples.
*NPS= No Parasites Seen; Error bars denote 95% confidence intervals.
| Raw count of sampled dogs by descriptive characteristic and parasitism status* | |||
| Parasitized | Total count | ||
| Yes | No | ||
| Age category | |||
| Pediatric (< 6 months) | 116 | 30 | 146 |
| Juvenile (6 months – 1 year) | 58 | 18 | 76 |
| Adult (1 year – 7 years) | 199 | 80 | 279 |
| Senior (> 7 years) | 7 | 8 | 15 |
| Unknown | 4 | 2 | 6 |
| Sex | |||
| Male | 193 | 58 | 251 |
| Female | 190 | 80 | 270 |
| Unknown | 1 | 0 | 1 |
| Altered status | |||
| Intact | 360 | 113 | 473 |
| Altered | 21 | 24 | 45 |
| Unknown | 3 | 1 | 4 |
| Intake source | |||
| Stray | 321 | 115 | 436 |
| Owner surrender | 32 | 13 | 45 |
| Transfer | 4 | 2 | 6 |
| TNR | 20 | 3 | 23 |
| Unknown | 7 | 5 | 12 |
| *Counts indicate presence of at least one of the common gastrointestinal parasites evaluated in this study (presumed Ancylostoma spp., Toxocara spp., Trichuris spp. and Cystoisospora spp). | |||
As with cats, logistic regression was used to evaluate risk factors for parasitism. Age was associated with overall parasitism (P = 0.035). Pediatric dogs had 1.6 times greater odds of parasitism versus adults (P = 0.014), and senior dogs had 0.35 lesser odds of parasitism versus adults (P = 0.016). In addition, pediatric dogs had 4.4 times the odds of parasitism compared to senior animals (P = 0.014). Further analysis revealed that pediatric dogs had 0.26 times lesser odds (P = 0.0005), and juvenile dogs had 1.4 greater odds (P = 0.014) of Trichuris spp. versus adult dogs. An age association was demonstrated for both Toxocara spp. and Cystoisospora spp. infections (P ≤ 0.0001, P ≤ 0.0007), but differences between age categories were not detected; as mentioned with intake type and cats, this is likely due to small sample size after partitioning of age categories (Table 3). The interaction of intact status and sex was also associated with overall parasitism and Ancylostoma spp. infections. Intact male dogs had 2.5 times greater odds of overall parasitism compared to other sex classifications (P ≤ 0.0001), and they had 2.1 times greater odds of Ancylostoma spp. compared to other sex classifications (P = 0.0009). No other significant interactions were observed.
| Raw count and percentage of sampled dogs by age category* | ||||
| Age category+ | ||||
| Toxocara spp. (%) | Cystoisospora spp. (%) | |||
| Yes | No | Yes | No | |
| Pediatric | 60 (64.0) | 86 (20.4) | 21 (80.8) | 130 (26.3) |
| Juvenile | 13 (13.8) | 63 (15.0) | 1 (3.8) | 75 (15.2) |
| Adult | 21 (22.3) | 258 (61.1) | 4 (15.4) | 275 (55.6) |
| Senior | 0 (0.0) | 15 (3.6) | 0 (0.0) | 15 (3.0) |
| Total | 94 | 422 | 26 | 495 |
| *Counts are not exclusive between parasite categories – animals may have been positive for multiple parasites. +Animals in which age was not determined are not included. |
||||
Discussion
Feline
The sample size for cats was lower than desired due to sampling limitations; many housing units contained more than three cats. Therefore, determining which environmental sample originated from which patient was not possible. Arguably, a representative sample could have been obtained, but the authors wished to maintain the integrity of patient-sample alignment and avoid evaluation of clusters. In addition, to reduce handling stress and promote overall physical health of the shelters’ animals, fecal loop sampling in cats was restricted. Whenever possible, environmental sampling was performed.
Prevalence results were similar to a previously mentioned regional study evaluating shelter cats,4 with approximately half of this study’s cats being diagnosed with at least one parasite on intake to Mississippi shelters. It is likely that similar climate, environmental terrain, and parasite survival within the southeastern region of the United States would support similar parasite burdens for shelter populations in differing states. Coinfections were uncommon, which could be a factor of diagnostic test, sample procurement, low regional prevalence, or another undetermined contributor. Furthermore, within this study, a cat’s intake source played a significant role in odds of overall parasitism; presenting to the shelter as a stray intake was identified as a risk factor for parasitism.
When evaluating the raw count of cats enrolled (Table 4), strays comprised the largest number of study animals, which is both representative of many shelter populations and potentially contributing to the results. Other potential contributors to this finding could be sheer intake numbers coupled with exposure and population density relationships. Many gastrointestinal parasites survive and develop in soil, greatly increasing environmental exposure of outdoor cats to parasites. In the United States, a large portion of owned cats are indoor only.20 Indoor cats may be more likely to have external identification or be microchipped; therefore, they are plausibly less commonly picked up and taken to shelters as strays. However, owned cats that are allowed outside or lack identification may reasonably account for some of the stray intakes at a shelter. Owned cats are reliant on owner maintenance of environmental cleanliness and provision of preventive care. Indoor cats may be more likely to induce direct transmission via shared litterboxes and allogrooming, but the overall benefits of ownership seemingly outweigh the incidence of transmission within multi-cat households. Within this study, there was no ability to determine which cats may have been surrendered from multi-cat or indoor/outdoor cat households. It is reasonable to assume that the majority of stray intakes are truly unowned animals or may come from households that elected not to provide preventive care or implement identification such as a microchip or collar. Regardless, a cat’s intake type should be a factor to consider for empirical clinical decisions.
| Raw count and percentage of sampled cats by intake type and parasitism status* | |||
| Intake type | |||
| Parasitized (%) | Total | ||
| Yes | No | ||
| Owner surrender | 4 (3.3) | 15 (12.2) | 19 (15.4) |
| Stray | 50 (40.7) | 43 (35.0) | 93 (75.6) |
| Transfer | 3 (2.4) | 4 (3.3) | 7 (5.7) |
| Total | 59 (48.0) | 64 (52.0) | 123 |
| *Counts indicate presence of at least one of the common gastrointestinal parasites evaluated in this study (presumed Ancylostoma spp., Toxocara spp., Trichuris spp. and Cystoisospora spp). | |||
Lastly, juvenile cats (6 months of age to 1 year of age) were found to have greater odds of having Ancylostoma spp. over adult or pediatric cats. This finding is also difficult to relate to a specific cause. The increased odds may be due to complicating factors such as prepatent periods in young cats or an undetermined age resistance to parasitism in adults. Alternatively, this finding may be due to behavioral differences in juvenile cats. Juveniles have surpassed most development thresholds of kittens (teeth eruption, nursing, walking, litterbox usage, etc.) and are actively exploring the world around them during this life stage. Juveniles explore various socialization interactions such as grooming, scratching, and territorial marking. These particular behaviors place this life stage at greater odds of contracting parasites; though adults carry these behaviors throughout their lives, they may be less likely to seek social interaction versus juvenile cats. Other explanations for this finding potentially include sampling challenges. Environmental sampling is susceptible to contamination (fur, food, other bodily secretions, litter), but fecal loop sampling obtains smaller aliquots of feces for testing compared to environmental sampling. Regardless of sampling method, pediatric patients produce smaller fecal sample size. Temperament difficulties are often more common in adults. These challenges result in lower diagnostic sensitivity and potentially false negative results. Lastly, results from partitioned groups with small sample sizes (i.e. pediatric and senior animals) should be interpreted cautiously; limited samples may hinder the accuracy of reported results.
Canine
Prevalence of gastrointestinal parasites in dogs enrolled in this study was higher than previous regional reports.3,7,21 The authors suggest that this study demonstrates increased prevalence due to obtaining samples at intake. Other studies sampled dogs already housed in the shelter, and potentially, these patients may have already been administered antiparasitic therapies. One study7 that focused on intake sampling reported a prevalence of 54% for any gastrointestinal parasite at intake over a short period of time (June–July 2017). Utilizing dogs that have undergone intake procedures and a short time-frame for sampling plausibly limit the ability to extrapolate previous study data to other shelters. Data collected from multiple shelters over an extended period (approximately 2 years) in this study, resulting in an overall prevalence of 74%, may be more representative of canine intake populations for the southeastern region. Ancylostoma spp. were the most commonly diagnosed parasite, and coinfections were common in dogs, with Ancylostoma spp. and Trichuris spp. being the most frequent coinfection. Of all enrolled dogs, a considerable percentage (68%) presented with either Ancylostoma spp. or Toxocara spp. infections, posing a zoonotic risk to humans. These data suggest that dogs in Mississippi and potentially within the southeastern region of the United States are likely to present with a zoonotic parasite.
Age was found to be significantly associated with overall parasitism and various specific parasites. Younger and older age categories may have differing odds of parasitism due to immune function in these life stages. Young animals are developing immunocompetency, while older animals have established immunocompetency in most cases. Specifically with young age categories, hygienic behaviors are less common. Puppies reputationally engage in coprophagia, teething on fomites, and other curious investigation of their surroundings – all behaviors put them at greater odds of gastrointestinal parasite transmission from both littermates and the environment. An age-associated risk for both Toxocara spp. and Cystoisospora spp. was observed within this study, but infrequent identification of these particular parasites prevented determination of high-risk age categories. However, speculation from previous studies3,5,6,21 and other results in this study suggest that younger age groups are the higher risk categories.
Neuter status and sex were significantly associated with overall parasitism, a finding that has been minimally reported and discussed in other literature.3,6 In agreement with the few reports from previous literature, intact male dogs had greater odds of overall parasitism over other sex classifications. Additionally, intact male dogs had greater odds of hookworm infections compared to other sex classifications, a finding that is reasonably explained by the high prevalence of Ancylostoma spp. given in this study. Intact males tend to engage in more ‘risky’ behaviors such as roaming, territorial aggression, and environmental investigation that other sex classifications less commonly engage in. These behaviors may increase the risk for parasitism in this sex classification; however, hormonal influence on the immune system and the animal’s ability to prevent infection altogether cannot be ruled out. As is mentioned in other literature,3,6,17 encysted larvae and larval migration are parasitic behaviors more likely to affect intact females versus spayed females; therefore, other interactions related to sex hormones and the immune system may be undetermined. Conclusions regarding hormonal influence cannot be made with the present data in this study. Another explanation is that surgical alteration may indicate that an animal is more likely to have received routine veterinary care, including preventive antiparasitics.6 If this is the case for animals entering shelters, parasite prevalence in surgically altered animals is more likely to align with the lower prevalences reported in pet dogs.
Lastly, it must be acknowledged that there is the chance of misclassification of animals entering the shelter system. If a roaming animal is confiscated by an animal control entity or brought into the shelter by a good Samaritan, it cannot be definitively determined that the animal is a stray. Animals with no identification (collar, tags, microchip, etc.) are most often classified as stray animals. A majority of the samples obtained in this study were sourced from stray animals, therefore representing a large portion of the overall evaluated shelter population. Certainly, a portion of these stray animals are simply ‘temporarily missing’, but there is no method for determining at intake, which animals are truly awaiting owner reclaim. A lack of knowledge about an animal entering the shelter system is unfortunately a common challenge for staff caring for these animals, and decisions must be made regarding care with the information available. This particular factor likely contributes to the findings for both dogs and cats, though to which degree misclassification plays a role cannot be verified.
Conclusion
Regardless of challenges and unclear associations, the data present useful findings for developing anthelmintic protocols in shelters. Though just under half of cats entering Mississippi shelters presented with at least one parasite, it remains that just over half of the cats were not found to present with a gastrointestinal parasite. A considerably greater percentage of dogs presented with at least one parasite, but multiple associations with overall parasitism and particular parasites were observed. Knowledge of regional prevalence and factors placing patients at higher or lower risk enables shelters to make informed decisions regarding antiparasitic therapies and environmental decontamination.
It must be acknowledged that passive fecal floatation is an imperfect test for gastrointestinal parasite diagnosis; however, many shelters lack access to a centrifuge and may divert resources to other needs. In this study, the sites of sample procurement and sample evaluation lacked access to a centrifuge and financial constraints prevented obtainment. Centrifugation or sedimentation would improve procurement of parasite ova, but these methods require further training and resources. Passive floatation has less diagnostic sensitivity and creates opportunities for false negatives. Reliability of results would be strengthened with confirmatory testing through secondary diagnostics (polymerase chain reaction, Baermann, direct smear, etc.), but this would require additional time and finances that shelter staff may wish to allocate to other needs. Despite lower sensitivity, passive floatation is a common method used in many shelters due to affordability and limited need for staff training. The results of this particular method will often be used to guide point of care treatment decisions. Recognizing potential risk factors and that a portion of animals will not be ‘positive’ for parasites on intake, evaluation of each animal’s risk for specific parasites is beneficial to exercising judicious use of anthelmintic products and preserving shelter resources. Nevertheless, care must be taken to protect enzootic and zoonotic transmission, especially when diagnostic evaluation is not possible. Consideration for appropriate PPE and environmental decontamination must be included in staff training and routine shelter supplies. When environmental transmission can be prevented and staff are provided education to understand human and animal risks, more careful use of anthelmintic products can be implemented. The initiative to treat all patients is reliable only as long as treatment works; management perspectives should begin including other preventive measures to benefit future clinical and financial decisions.
The authors declare that they have no conflicts of interest to disclose. This work was supported by Mississippi State University College of Veterinary Medicine Office of Research and Graduate Studies House Officer Grant and the National Institute of General Medical Sciences of the National Institutes of Health (Award Number U54GM115428).
Acknowledgments
The authors would like to thank Drs. Alexis Thompson, Hannah Urig, and Keegan Jones for contributions to this work through critical technical support and guidance of study analysis.
References
| 1. | Empirical Definition & Meaning – Merriam-Webster. Accessed Feb 26, 2024. https://www.merriam-webster.com/dictionary/empirical |
| 2. | The Association of Shelter Veterinarians. The Guidelines for Standards of Care in Animal Shelters: Second Edition. JSMCAH. 2022;1(S1):1–76. doi: 10.56771/ASVguidelines.2022 |
| 3. | Blagburn B, Lindsay D, Vaughan J, et al. Prevalence of Canine Parasites Based on Fecal Floatation. Compend Contin Educ Pract Vet. 1996;18(5):483–509. |
| 4. | Hoggard KR, Jarriel DM, Bevelock TJ, Verocai GG. Prevalence Survey of Gastrointestinal and Respiratory Parasites of Shelter Cats in Northeastern Georgia, USA. Vet Parasitol Reg Stud Reports. 2019;16:100270. doi: 10.1016/j.vprsr.2019.100270 |
| 5. | Little SE, Johnson EM, Lewis D, et al. Prevalence of Intestinal Parasites in Pet Dogs in the United States. Vet Parasitol. 2009;166(1–2):144–152. doi: 10.1016/j.vetpar.2009.07.044 |
| 6. | Mohamed AS, Moore GE, Glickman LT. Prevalence of Intestinal Nematode Parasitism Among Pet Dogs in the United States (2003–2006). J Am Vet Med Assoc. 2009;234(5):631–637. doi: 10.2460/javma.234.5.631 |
| 7. | Campanale D, Walden H, Garcia L, Crawford C, Hernandez J. Zoonotic and Non-Zoonotic Intestinal Parasites in Shelter Dogs at Admission and Before. JSMCAH. 2023;2(1). doi: 10.56771/jsmcah.v2.9 |
| 8. | CAPC Parasite Prevalence Maps. Companion Animal Parasite Council. 2023. Accessed Feb 26, 2024. https://capcvet.org/ |
| 9. | Hookworms. Companion Animal Parasite Council. March 2023. Accessed Feb 26, 2024. https://capcvet.org/guidelines/hookworms/ |
| 10. | Ascarid. Companion Animal Parasite Council. September 2022. Accessed Feb 26, 2024. https://capcvet.org/guidelines/ascarid/ |
| 11. | Centers for Disease Control and Prevention. Epi Info 7 (Version 7.2.5). [Computer software]. 2008. Atlanta, GA, USA. https://cdc.gov/epiinfo/. |
| 12. | Dentistry – Veterinary Preventive Medicine. Accessed Jul 30, 2025. https://pressbooks.umn.edu/vetprevmed/chapter/chapter-8-dentistry/ |
| 13. | Aging Puppies by Teeth. Published online July 2023. Accessed Jul 30, 2025. https://www.aphis.usda.gov/sites/default/files/ac-aid-aging-puppies-by-teeth.pdf |
| 14. | Broussard JD. Optimal Fecal Assessment. Clin Tech Small Anim Pract. 2003;18(4):218–230. doi: 10.1016/S1096-2867(03)00076-8 |
| 15. | Zajac AM, Conboy GA. Veterinary Clinical Parasitology. 8th ed. Hoboken, New Jersey, USA: Wiley-Blackwell; 2012. |
| 16. | Bowman DD, Fogarty EA. Parasitology: Diagnostics in Dogs and Cats. The Gloyd Group; 2003, St. Louis, Missouri, USA: Nestle Purina Petcare Company. |
| 17. | Bowman DD, ed. Georgis’ Parasitology for Veterinarians. 11th ed. Elsevier; 2021. |
| 18. | Microsoft Corporation. Microsoft Excel [Computer software]. 2018. Redmond, WA. https://office.microsoft.com/excel. |
| 19. | SAS Institute Inc. Statistical Analysis Software (Version 9.4) [Computer software]. 2013. Cary, NC, USA. https://sas.com/en_us/software/stat.html. |
| 20. | Grigg EK, Kogan LR. Owners’ Attitudes, Knowledge, and Care Practices: Exploring the Implications for Domestic Cat Behavior and Welfare in the Home. Animals. 2019;9(11):978. doi: 10.3390/ani9110978 |
| 21. | Raza A, Rand J, Qamar AG, Jabbar A, Kopp S. Gastrointestinal Parasites in Shelter Dogs: Occurrence, Pathology, Treatment and Risk to Shelter Workers. Animals (Basel). 2018;8(7):108. doi: 10.3390/ani8070108 |